Vitamin C (Ascorbic Acid)
Vitamin C refers to L-ascorbic acid in humans, whereas its stereoisomer D-ascorbic acid is biologically inactive. It is a water-soluble, white, crystalline, odorless compound with a characteristic sour taste. Chemically, it is a strong reducing agent and is therefore highly susceptible to oxidation.
Ascorbic acid is readily destroyed by oxidative processes and exposure to atmospheric oxygen, particularly in the presence of catalytic metal ions such as iron (Fe) and copper (Cu). It is also unstable in alkaline conditions, as alkalis promote its rapid degradation, whereas acidic environments help preserve it. For this reason, vitamin C is better preserved in acidic fruits than in alkaline media.
From a nutritional and biochemical standpoint, ascorbic acid is not required in the diet of most animals because they are capable of synthesizing it endogenously from glucose via the uronic acid pathway. However, humans, other primates, guinea pigs, and a few other species lack the enzyme L-gulonolactone oxidase, which is essential for the final step of this pathway. Consequently, these species are unable to synthesize vitamin C and must obtain it from dietary sources, making it an essential vitamin for them.
 Absorption, Distribution, and Fate in the Body
Absorption, Distribution, and Fate in the Body
Ascorbic acid is absorbed mainly in the small intestine, primarily in the jejunum, through sodium-dependent active transport mechanisms mediated by SVCT1 and SVCT2 (sodium-dependent vitamin C transporters). At lower to moderate dietary concentrations, absorption is highly efficient; however, at higher doses, absorption becomes saturated and excess is excreted.
After absorption, vitamin C is widely distributed throughout the body. Relatively high concentrations are found in metabolically active and endocrine-rich tissues such as the adrenal glands, pituitary gland, leukocytes, and retina, reflecting its role in enzymatic and protective functions.
The body’s storage capacity for vitamin C is limited, and it is not stored in large reserves like fat-soluble vitamins. Any excess intake beyond tissue saturation is excreted in urine, primarily as unmetabolized ascorbic acid and its oxidation products.
 Occurrence of Vitamin C
Occurrence of Vitamin C
Fresh fruits and vegetables are the most important dietary sources of vitamin C, whereas animal tissues are generally poor sources. Among plant foods, citrus fruits and tomatoes are considered the richest and most reliable sources. Other significant sources include green peppers, onions, spinach, cabbage, turnips, melons, and potatoes.
Milk contains relatively low amounts of vitamin C; however, human milk contains higher concentrations (5–7 mg/100 mL) compared to cow’s milk (1.0–2.6 mg/100 mL), although both are considered limited sources.
Orange juice is a practical dietary source, with approximately 25 mg of ascorbic acid per ounce, which contributes significantly toward the nutritional requirement of infants, whose estimated daily requirement is about 40 mg/day in early infancy.
Vitamin C is highly labile during food processing. It is easily destroyed by heat, prolonged cooking, boiling in water, and exposure to air, as these conditions accelerate oxidation. Therefore, raw or lightly cooked foods retain significantly higher vitamin C content compared to overcooked foods.
 Changes Undergone by Vitamin C in the Body
Changes Undergone by Vitamin C in the Body
In biological systems, ascorbic acid undergoes reversible oxidation-reduction reactions. It is first oxidized to dehydroascorbic acid, which retains full vitamin activity. Dehydroascorbic acid can further undergo irreversible degradation to 2,3-diketogulonic acid, and eventually to oxalic acid as an end product. These reactions are shown below:
Ascorbic acid ⇌ Dehydroascorbic acid → 2,3-diketogulonic acid → Oxalic acid
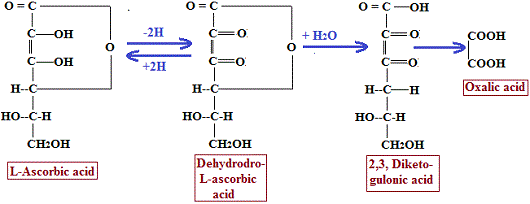
The reversible interconversion between ascorbic acid and dehydroascorbic acid is of major physiological significance. This property enables vitamin C to function as an important participant in cellular oxidation-reduction (redox) reactions, where it can readily donate and accept electrons.
 Biochemical Functions in the Body
Biochemical Functions in the Body
In addition to its role as a redox agent, vitamin C serves as an essential cofactor for several hydroxylation reactions. One of its most critical roles is in the post-translational modification of collagen, where it acts as a cofactor for prolyl and lysyl hydroxylases. Hydroxylation of proline and lysine residues is essential for stabilizing the triple-helical structure of collagen, thereby maintaining the integrity of connective tissue, blood vessels, bone matrix, and skin. Impaired collagen synthesis directly explains many features of vitamin C deficiency.
Vitamin C is also required for the synthesis of carnitine, which plays a key role in the transport of long-chain fatty acids into mitochondria for β-oxidation. In addition, it participates in the biosynthesis of certain neurotransmitters, particularly norepinephrine, by acting as a cofactor in dopamine β-hydroxylase-mediated reactions.
Another important function of vitamin C is its role in enhancing intestinal absorption of non-heme iron. It facilitates this process by reducing ferric iron (Fe³⁺) to the more absorbable ferrous form (Fe²⁺), thereby improving iron bioavailability and preventing iron deficiency.
 Deficiency Manifestations
Deficiency Manifestations
Deficiency of vitamin C leads to scurvy, a disease primarily resulting from defective collagen synthesis. Clinical features include bleeding gums, loosening of teeth, delayed wound healing, petechial and perifollicular hemorrhages, weakness, and impaired bone formation. These manifestations reflect the structural weakness of connective tissues due to inadequate hydroxylation of collagen fibers.




