Vitamin D – Structure and Occurrence
Vitamin D is a fat-soluble secosteroid prohormone, meaning it is derived from a steroid structure in which one of the rings (the B-ring) is broken. This unique structural modification distinguishes it from classical steroid hormones and is essential for its biological activity after activation in the body.
 Structure of Vitamin D
Structure of Vitamin D
Many compounds possess anti-rickets activity, but only two naturally occurring forms are recognized as true vitamin D: vitamin D₂ and vitamin D₃.
Vitamin D₂, now called ergocalciferol, is formed by ultraviolet irradiation of ergosterol, a sterol originally isolated from ergot (a fungus). Because ergosterol is widely present in plants and fungi, vitamin D₂ is considered to be of plant/fungal origin. Structurally, ergocalciferol differs from vitamin D₃ by the presence of a double bond and an additional methyl group in its side chain, which slightly alters its biological potency and metabolism.
Vitamin D₃, now called cholecalciferol, is of animal origin and is also synthesized in human skin. It is formed when 7-dehydrocholesterol (provitamin D₃) in the epidermis is exposed to ultraviolet light. UVB radiation (wavelength 290–315 nm) initiates a non-enzymatic photochemical reaction, in which B-ring cleavage of the steroid nucleus occurs, producing previtamin D₃, which then undergoes thermal isomerization to form vitamin D₃.
The reaction occurs mainly in the epidermis, particularly in the stratum basale and stratum spinosum, where 7-dehydrocholesterol is present in high concentration.
The formation of ergocalciferol and cholecalciferol from their precursors is shown below:
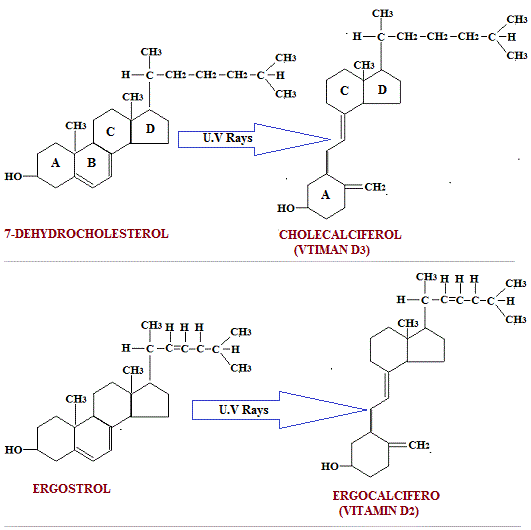
The reaction, ergsterol àergocalciferol is not 100% complete. Other compounds are also formed, e.g. lumisterol and tachysterol both of which are devoid of vitamin D activity. Tachysterol on hydrogenation is converted to dihydrotachysterol or A.T. 10 D3 have same physiological actions in man. In nature they occur as esters.
 Regulation of Cutaneous Synthesis
Regulation of Cutaneous Synthesis
Cutaneous synthesis of vitamin D is influenced by several physiological and environmental factors.
- Melanin acts as a natural sunscreen by absorbing UVB radiation and is the most important biological inhibitor of vitamin D synthesis.
- Keratin provides a minor physical barrier.
- Therefore, individuals with darker skin pigmentation require longer sunlight exposure to produce equivalent vitamin D levels.
- Aging reduces 7-dehydrocholesterol content in skin, decreasing synthesis.
- Obesity reduces bioavailability due to sequestration in adipose tissue.
- Environmental factors such as latitude, season, time of day, clothing, sunscreen use, and air pollution significantly influence production.
Importantly, UVB radiation is completely blocked by ordinary window glass and significantly reduced by atmospheric pollution, smoke, and dust.
 Photochemical Regulation and Photoproducts
Photochemical Regulation and Photoproducts
The formation of vitamin D from ergosterol and 7-dehydrocholesterol is not fully efficient. Several photoproducts are formed simultaneously, including:
- Lumisterol
- Tachysterol
These compounds are biologically inactive and form part of a natural regulatory mechanism called the photostationary system, which prevents excessive vitamin D production during prolonged sunlight exposure by shifting photochemical equilibrium.
Tachysterol can be hydrogenated to form dihydrotachysterol (AT-10 D₃), a synthetic analog with vitamin D–like activity, particularly in calcium metabolism.
 Transport and Activation
Transport and Activation
Vitamin D is a lipid-soluble molecule and does not circulate freely in plasma. After synthesis in skin or absorption from diet, it binds to vitamin D–binding protein (DBP) for transport to the liver.
Vitamin D exists in biologically inactive forms and requires two hydroxylation steps for activation:
- Liver: Conversion to 25-hydroxyvitamin D (calcidiol)
- Kidney (proximal tubules): Conversion to 1,25-dihydroxyvitamin D (calcitriol)
Calcitriol is the biologically active form responsible for most physiological actions.
 Endocrine Regulation of Vitamin D Metabolism
Endocrine Regulation of Vitamin D Metabolism
Vitamin D activation is tightly regulated by endocrine signals:
- Parathyroid hormone (PTH): stimulates renal 1α-hydroxylase → increases calcitriol
- Low serum calcium: indirectly increases calcitriol via PTH
- High phosphate levels: stimulate FGF-23
- Fibroblast growth factor-23 (FGF-23): inhibits calcitriol synthesis and promotes phosphate excretion
This integrated axis maintains calcium and phosphate balance within narrow physiological limits.
Calcitriol also participates in negative feedback inhibition of PTH, forming a self-regulating endocrine loop.
 Vitamin D Receptor (VDR) and Genomic Action
Vitamin D Receptor (VDR) and Genomic Action
The biological actions of vitamin D are mediated through the vitamin D receptor (VDR), a nuclear receptor present in target tissues such as intestine, bone, kidney, and immune cells.
Calcitriol binds to VDR and regulates gene transcription, leading to synthesis of proteins involved in calcium transport, bone remodeling, and immune regulation. This explains why vitamin D is now considered a hormone rather than a simple vitamin.
 Storage, Distribution, and Half-life
Storage, Distribution, and Half-life
Vitamin D is stored primarily in:
- Adipose tissue
- Liver (as 25-hydroxyvitamin D)
Because of its fat solubility, it has a long biological half-life, allowing maintenance of plasma levels over extended periods even with intermittent intake or sunlight exposure.
 Occurrence of Vitamin D
Occurrence of Vitamin D
Vitamin D is much less widely distributed in nature compared to many other vitamins.
Animal sources (Vitamin D₃):
- Cod liver oil (richest source)
- Fatty fish (salmon, mackerel, sardines)
- Egg yolk
- Limited amounts in liver and milk
Plant/fungal sources (Vitamin D₂):
- UV-exposed mushrooms (ergosterol → ergocalciferol)
- Yeast and fungi after irradiation
Human breast milk contains relatively low vitamin D unless maternal status is adequate or supplementation is provided.
Milk and eggs contain variable amounts depending on animal diet and sunlight exposure.
 Food Fortification and Modern Sources
Food Fortification and Modern Sources
Modern nutrition utilizes UV irradiation and fortification techniques to increase vitamin D content in foods, especially:
- Milk products
- Mushrooms
- Cereals and fortified foods
This compensates for limited natural dietary sources.
 Vitamin D as a Hormone Precursor
Vitamin D as a Hormone Precursor
It is now well established that vitamin D is not merely a dietary vitamin but a hormone precursor synthesized in the skin under environmental regulation. Its activation is tightly controlled by endocrine systems involving the kidney, parathyroid glands, and liver.
 Clinical Correlation (Brief Overview)
Clinical Correlation (Brief Overview)
Defects in vitamin D synthesis or action lead to:
- Rickets (children): defective mineralization of growing bone
- Osteomalacia (adults): defective mineralization of mature bone
These conditions reflect failure of calcium–phosphate homeostasis due to inadequate calcitriol activity.
 Integrated Concept
Integrated Concept
Vitamin D functions as a sunlight-dependent endocrine regulator that integrates environmental exposure, dietary intake, and internal metabolic pathways to maintain skeletal integrity and mineral balance. Its biological importance lies not only in bone health but also in gene regulation, immune modulation, and systemic calcium–phosphate homeostasis.




