Vitamin B2 (Riboflavin)
Chemical Structure
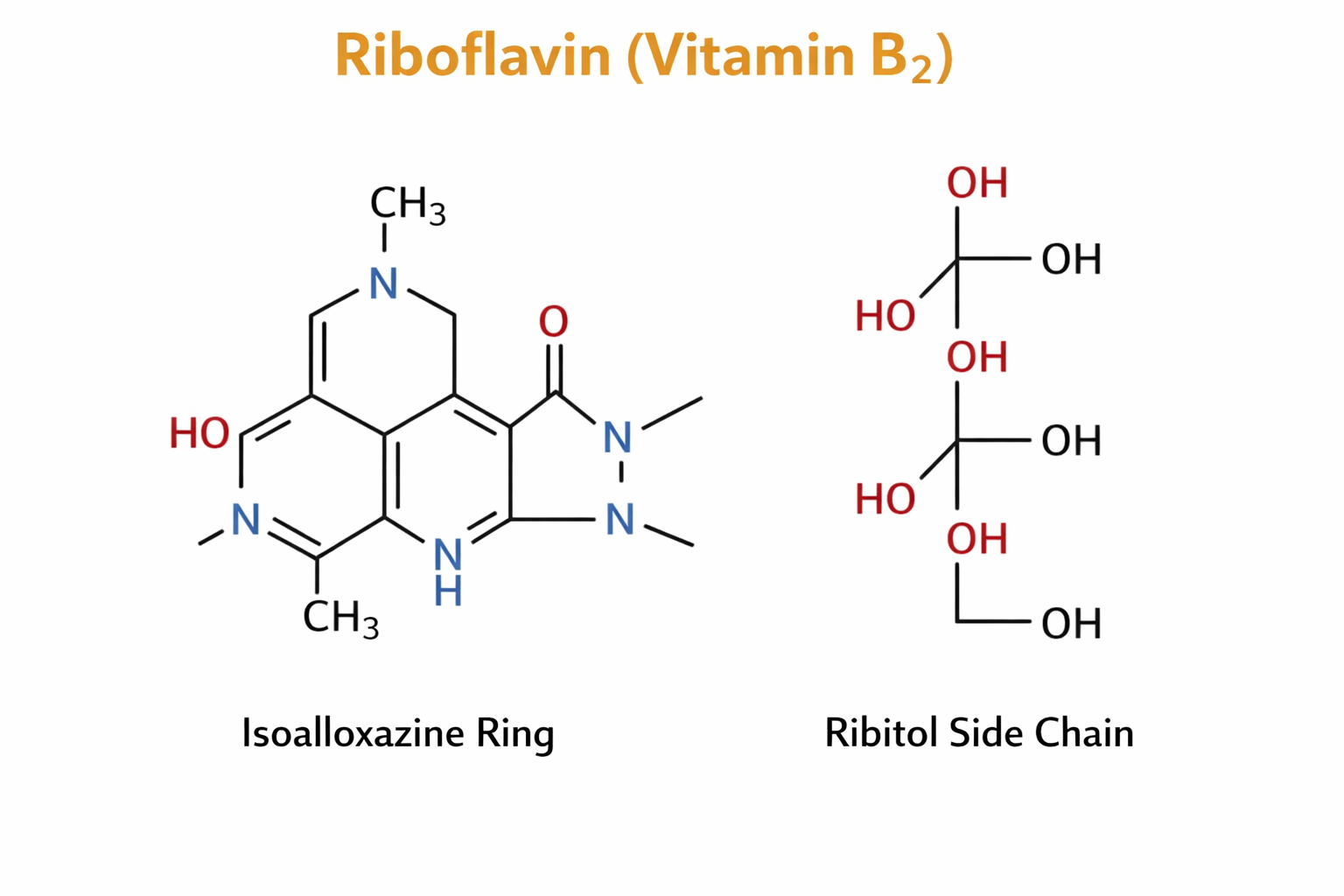
Riboflavin is a yellow, crystalline, water-soluble compound composed of a heterocyclic isoalloxazine ring attached to a sugar alcohol side chain known as ribitol. The name “riboflavin” is derived from “ribose” (reflecting the ribitol moiety) and “flavin” (from flavus, meaning yellow).
Riboflavin: Isoalloxazine ring − Ribitol side chain
The isoalloxazine ring system is the functional core of the molecule and is responsible for its redox properties. This ring can undergo reversible reduction and oxidation, allowing riboflavin to participate effectively in electron transfer reactions.
Structurally, riboflavin consists of:
- A tricyclic isoalloxazine ring, which accepts and donates electrons
- A ribitol side chain, which enhances solubility and facilitates enzymatic binding
The coenzyme derivatives of riboflavin are formed by modification of this structure:
- Flavin mononucleotide (FMN): formed by phosphorylation of riboflavin
- Flavin adenine dinucleotide (FAD): formed by addition of an adenosine monophosphate (AMP) moiety to FMN
These structural modifications enable tight binding to specific enzymes as prosthetic groups.
Physical Properties
- Riboflavin is a yellow–orange crystalline substance that gives a yellow color to solutions and urine.
- It is slightly soluble in water, and solubility increases with heat.
- It is insoluble in nonpolar solvents like chloroform and ether.
- It shows yellow-green fluorescence under ultraviolet (UV) light.
Chemical Properties
- Riboflavin is heat-stable, so it is not easily destroyed during cooking.
- It is very sensitive to light, especially UV light, and can be destroyed when exposed (e.g., milk in clear bottles).
- It is stable in acidic conditions but unstable in alkaline conditions.
- It acts as a redox compound, meaning it can accept and donate electrons in metabolic reactions.
 Occurrence of Vitamin B₂
Occurrence of Vitamin B₂
Vitamin B₂, commonly known as riboflavin, occurs in biological systems either as free riboflavin or as a component of complex proteins known as flavoproteins, which function predominantly as enzymes. It is widely distributed in nature and is present in virtually all living cells of both plant and animal origin.
Rich dietary sources of riboflavin include liver, wheat germ, yeast, milk, meat, fish, and eggs. Leafy green vegetables, fruits, and most root vegetables also contribute appreciable amounts. Although grains and cereals are relatively poor sources, their riboflavin content increases during germination. Dietary deficiency is frequently associated with low consumption of milk and animal-derived proteins.
Riboflavin is relatively stable to heat; however, it is highly sensitive to ultraviolet and visible light. Consequently, significant losses may occur when foods—particularly milk—are stored in transparent containers and exposed to light. Pasteurization of milk can also result in a measurable reduction in riboflavin content.
In contemporary nutrition, food fortification has become an important additional source of riboflavin. Fortified cereals, enriched flour, and certain beverages contribute significantly to intake in many populations. Although limited synthesis of riboflavin by intestinal microbiota has been described, it is not considered a reliable source for human nutritional needs. Being a water-soluble vitamin, riboflavin is susceptible to loss during cooking, especially with boiling and prolonged exposure to water.
 Biochemical Role of Vitamin B₂
Biochemical Role of Vitamin B₂
Riboflavin serves as a precursor for two essential coenzymes:
FMN (Flavin Mononucleotide)
FAD (Flavin Adenine Dinucleotide)
These coenzymes are integral components of a wide variety of oxidation–reduction (redox) reactions in cellular metabolism.
FMN functions as a coenzyme for several enzymes, including cytochrome c reductase, L-amino acid dehydrogenase, and Warburg’s yellow enzyme. FAD serves as a coenzyme for enzymes such as xanthine oxidase, liver aldehyde oxidase, and acyl-CoA dehydrogenase.
Enzymes that contain riboflavin-derived coenzymes are collectively termed flavoproteins. These enzymes play a central role in catalyzing redox reactions essential for cellular energy production.
In modern biochemical understanding, FMN and FAD act as tightly bound prosthetic groups in numerous mitochondrial dehydrogenases. They are critically involved in the electron transport chain, particularly within Complex I (FMN-dependent) and Complex II (FAD-dependent), thereby contributing directly to adenosine triphosphate (ATP) generation.
Beyond energy metabolism, riboflavin-dependent enzymes participate in multiple metabolic pathways, including:
- Fatty acid β-oxidation
- Amino acid metabolism, including oxidative deamination
- Conversion of tryptophan to niacin
Additionally, riboflavin plays an indirect but vital role in antioxidant defense. The FAD-dependent enzyme glutathione reductase maintains intracellular levels of reduced glutathione (GSH), which protects cells against oxidative stress and membrane damage.
 Deficiency of Vitamin B₂ (Ariboflavinosis)
Deficiency of Vitamin B₂ (Ariboflavinosis)
Riboflavin deficiency does not produce a single, distinct disease entity but instead manifests as a constellation of clinical features collectively referred to as ariboflavinosis. Tissues with high cellular turnover—particularly ectodermal tissues such as the skin, eyes, and mucous membranes—are most susceptible. Experimental Findings
Experimental Findings
In experimental animal models, particularly rats, riboflavin deficiency results in growth retardation, degeneration of nervous tissue, and ocular abnormalities such as corneal vascularization and cataract formation. Additional findings include dermatitis and alopecia. During pregnancy, deficiency may lead to congenital malformations in the developing embryo. Clinical Features in Humans
Clinical Features in Humans
In humans, riboflavin deficiency presents with characteristic mucocutaneous and ocular manifestations:
- Oral and Cutaneous Lesions:
- Inflammation and cracking of lips (cheilosis)
- Fissures at the angles of the mouth (angular cheilitis)
- Seborrheic dermatitis-like lesions, especially around the nose, ears, and nasolabial folds
- Greasy scaling of the skin
- Tongue Changes:
- Magenta-colored tongue (magenta glossitis)
- Atrophy and flattening of lingual papillae
- Ocular Manifestations:
- Conjunctival and corneal inflammation
- Corneal vascularization
- Photophobia (light sensitivity)
- Burning, itching, and dryness of the eyes
- Reduced visual acuity
These ocular symptoms are attributed to impaired flavoprotein-dependent oxidative metabolism in rapidly dividing epithelial tissues of the eye.
 Risk Factors and Predisposing Conditions
Risk Factors and Predisposing Conditions
Certain populations are at increased risk for riboflavin deficiency, including:
- Individuals with chronic alcoholism
- Patients with malabsorption syndromes (e.g., Celiac disease)
- Individuals with prolonged inadequate dietary intake
- Pregnant and lactating women with poor nutritional status
- Populations consuming predominantly refined, non-fortified cereal-based diets
Additionally, some medications, including phenothiazines and tricyclic antidepressants, may interfere with riboflavin metabolism and utilization.




