Vitamin K
 Occurrence of Vitamin K
Occurrence of Vitamin K
The best dietary sources of Vitamin K are spinach, cabbage, cauliflower and other green vegetables. Less efficient sources are tomatoes, cheese, egg yolk and liver. The normal bacterial flora, especially Gram-positive bacteria of the intestinal tract, contribute significant amounts of vitamin K2 (menaquinones).
Vitamin K is widely distributed in nature, particularly in green leafy vegetables where it is associated with chloroplast membranes. The dietary form predominantly present is phylloquinone (vitamin K1), whereas menaquinones (vitamin K2) are largely produced by intestinal microbiota and are also found in fermented foods and certain animal tissues.
 Absorption of Vitamin K
Absorption of Vitamin K
Bile salts are needed for its absorption and incorporation into micelles in the intestine for transport. After absorption it is stored only to a limited extent in the liver. Being fat soluble, its absorption is markedly reduced in conditions of fat malabsorption, obstructive jaundice and in prolonged use of antibiotics which disturb intestinal flora.
Vitamin K is absorbed in the small intestine along with dietary fats through a process dependent on bile salts and pancreatic secretions. It is incorporated into mixed micelles, taken up by enterocytes, and then packaged into chylomicrons for lymphatic transport. Because of this dependence on lipid absorption mechanisms, any condition that interferes with fat digestion or bile secretion significantly reduces its bioavailability.
Hepatic storage of vitamin K is minimal, and therefore continuous dietary intake and/or microbial synthesis are important for maintaining adequate levels.
 Description of Vitamin K
Description of Vitamin K
The name vitamin K stands for Koagulation vitamin given by Dam, a Danish worker, to a substance that prevented haemorrhagic tendency. It is a fat soluble and heat stable vitamin but is sensitive to light and oxidizing agents. There are 2 types of naturally occurring vitamin K. Vitamin K1 is obtained from plants such as alfalfa leaves while vitamin K2 is derived from bacterial synthesis. Both are derivatives of naphthoquinone and have the same general activity and both have been synthesized. Vitamin K1 is 2-methyl, 3-phytyl, 1,4-naphthoquinone and is termed phylloquinone. It has the following formula
Vitamin K is a group of structurally related fat-soluble compounds based on the naphthoquinone nucleus. It is chemically stable to heat but labile to light, alkali, and strong oxidizing conditions.
Phylloquinone (vitamin K1) is the principal dietary form and contains a phytyl side chain. It is biologically active and represents the major form found in green plants. It is not soluble in water but water soluble derivatives are available which are highly effective.
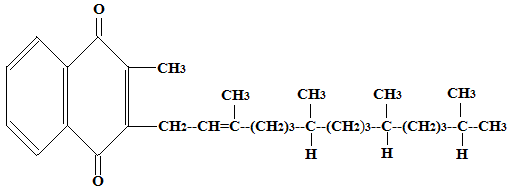
In vitamin K2 the side chain is different and contains isoprenoid units instead of the phytyl group of vitamin K1. It has the following general formula
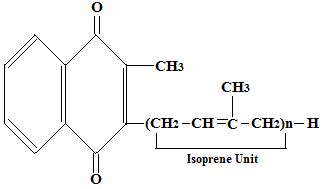
In the above formula, n (number of isoprenoid units) can range from 4 to 13 depending on the homolog (MK-4 to MK-13 are commonly found). Thus vitamin K2 consists of a group of compounds called menaquinones rather than only three forms. These menaquinones differ in the number of isoprenoid units in their side chains. MK-4 is found in animal tissues and is also formed from conversion of phylloquinone, while longer chain menaquinones (MK-7 to MK-13) are mainly produced by intestinal bacteria.
Menaquinones (vitamin K2) show variable chain length-dependent absorption and tissue distribution. Short-chain forms such as MK-4 are more widely distributed in animal tissues, whereas long-chain menaquinones tend to have longer half-lives and may contribute significantly to extrahepatic vitamin K functions.
Many vitamers are also available. One of these, menadione or vitamin K3, on a weight basis is more potent than naturally occurring vitamin K. It is oil soluble but its sodium bisulfite and sodium phosphate derivatives are water soluble and are used clinically both by mouth and by injection. However, these products may prove toxic and are in some cases less effective, e.g. in treating overdosage with dicumarol. Water soluble derivatives of vitamin K1 have also been obtained and have proved very effective.
Menadione is 2-methyl, 1,4-naphthoquinone and has the following formula
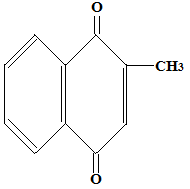
Menadione is a synthetic naphthoquinone derivative that can be converted in vivo to active vitamin K forms, although its clinical use is limited due to potential oxidative toxicity, particularly in high doses or in neonates.
 Vitamin K Function and Metabolism
Vitamin K Function and Metabolism
Vitamin K functions as an essential cofactor for the enzyme γ-glutamyl carboxylase, which catalyzes the post-translational carboxylation of glutamic acid residues to γ-carboxyglutamic acid (Gla) residues in vitamin K dependent proteins. These include clotting factors II (prothrombin), VII, IX, and X as well as anticoagulant proteins C and S. This reaction is essential for calcium binding and biological activity of these proteins.
This γ-carboxylation reaction occurs in the liver and is essential for converting inactive precursor proteins into biologically active clotting factors capable of binding calcium ions, which is necessary for their interaction with phospholipid surfaces during the coagulation cascade.
The active form of vitamin K is its reduced form, vitamin KH₂ (hydroquinone form). During the carboxylation reaction, vitamin KH₂ is oxidized to vitamin K epoxide, which is then recycled back to the active form through the vitamin K epoxide cycle. This recycling is essential for maintaining activity even with low dietary intake.
This regeneration process is known as the vitamin K cycle and ensures efficient reuse of vitamin K molecules. Without this cycle, continuous dietary intake would be required in much larger amounts.
The enzyme vitamin K epoxide reductase (VKOR) is responsible for regenerating active vitamin K. This enzyme is inhibited by dicumarol and warfarin, leading to reduced activation of clotting factors and anticoagulant effect.
Warfarin acts as a competitive inhibitor of VKOR, thereby depleting reduced vitamin K levels and producing a functional deficiency state that results in the synthesis of inactive clotting factors. This mechanism forms the basis of oral anticoagulant therapy.
Because vitamin K is stored only in small amounts in the liver and body reserves are limited, deficiency can develop relatively quickly in newborns, patients on prolonged antibiotic therapy, or in fat malabsorption syndromes.
A classic clinical condition associated with deficiency is hemorrhagic disease of the newborn, which occurs due to low placental transfer of vitamin K and sterile intestinal tract at birth. This condition is effectively prevented by prophylactic administration of vitamin K at birth.




