Vitamin B12 (Cyanocobalamin)
Vitamin B12 is the most potent of all vitamins and is also known as the anti-pernicious anemia vitamin due to its essential role in preventing and treating pernicious anemia. Historically, it was identified as the active “factor” in liver extracts responsible for the dramatic hematologic improvement seen in patients with this disease.
In earlier biochemical literature, vitamin B12 was referred to as the extrinsic factor or food factor of Castle, distinguishing it from the intrinsic gastric factor required for its absorption.
 Occurrence of Vitamin B12
Occurrence of Vitamin B12
Vitamin B12 is not synthesized by plants. Consequently, plant foods do not naturally contain this vitamin, although trace amounts may occasionally be present due to bacterial contamination or symbiotic bacterial activity on plant surfaces or within soil environments.
The richest natural sources of vitamin B12 are:
- Liver and kidneys (chief storage organs and richest dietary sources)
- Milk and dairy products
- Meat
- Eggs
- Fish
In humans, an additional potential source of vitamin B12 is the intestinal microbiota, although the amount produced is highly variable and generally insufficient to meet physiological requirements.
Thus, vitamin B12 in the human diet is fundamentally derived from animal-based foods, and its biological synthesis is carried out exclusively by microorganisms (bacteria).
 Absorption, Transport, and Storage of Vitamin B12
Absorption, Transport, and Storage of Vitamin B12
Vitamin B12 absorption is a highly specialized and tightly regulated process.
Absorption
- Vitamin B12 is absorbed only in the terminal ileum.
- Absorption requires the presence of intrinsic factor (IF), a glycoprotein secreted by gastric parietal cells.
- In the intestinal lumen, vitamin B12 binds intrinsic factor, forming a vitamin B12–intrinsic factor complex.
- This complex binds to specific ileal receptors known as cubilin receptors, enabling receptor-mediated endocytosis and absorption.
Transport
After absorption, vitamin B12 enters the bloodstream bound to specific transport proteins called transcobalamins:
- Transcobalamin II:
The biologically active transport protein responsible for delivering vitamin B12 to body tissues. - Transcobalamin I and III:
Primarily serve as binding and storage-related proteins in circulation.
Storage
- The liver is the principal storage organ for vitamin B12.
- It can store large reserves sufficient for several years.
- Because of this extensive hepatic storage, vitamin B12 deficiency typically develops slowly and may take years after reduced intake or absorption failure.
 Chemistry of Vitamin B12
Chemistry of Vitamin B12
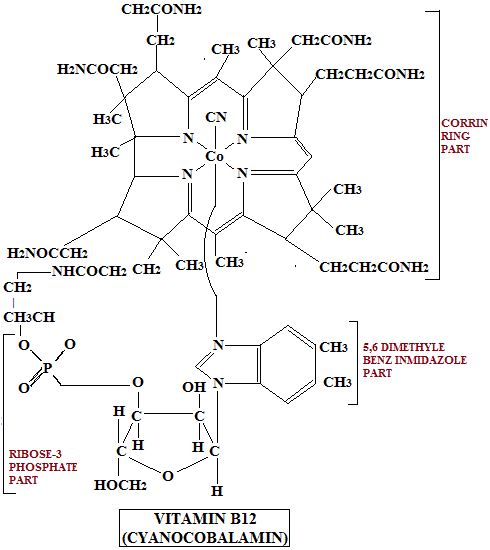
Vitamin B12 is unique among vitamins because it is the only naturally occurring organic compound containing cobalt.
Key chemical characteristics:
- Water-soluble vitamin
- Deep red, crystalline compound
- Tasteless
- Molecular weight: approximately 1355
- Stable at neutral pH, but unstable under alkaline conditions
Structural Features
The structure of vitamin B12 is complex and consists of multiple functionally important components.
There are several biologically active compounds collectively known as cobalamins, all sharing structural similarity.
Core structure:
- A central corrin ring, which resembles the tetrapyrrole structure of porphyrins but is less symmetrical.
- A single cobalt atom is positioned at the center of this ring.
- The cobalt atom can exist in variable oxidation states, which is essential for its biochemical activity.
The cobalt atom is:
- Coordinated to the four nitrogen atoms of the corrin ring
- Further coordinated to a nucleotide loop, which includes:
- 5,6-dimethylbenzimidazole
- Ribose
- Phosphate group
- Aminopropanol side chain attached to ring IV
Cobalt in vitamin B12 is not analogous to iron in hemoglobin. Iron (Fe²⁺/Fe³⁺) is present in heme, whereas cobalt is specific to vitamin B12 chemistry.
 Types of Vitamin B12 (Cobalamins)
Types of Vitamin B12 (Cobalamins)
Different forms of vitamin B12 arise based on the group attached to the cobalt atom. These variations often result from isolation or chemical processing rather than physiological diversity.
- B12 (Cyanocobalamin): contains CN group; most commonly used pharmaceutical form
- B12-a (Hydroxocobalamin): contains OH group; believed to have better retention in the body
- B12-b (Aquocobalamin): contains H₂O group
- B12-c (Nitrocobalamin): contains NO₂ group
- Additional forms: chlorocobalamin and thiocyanatocobalamin
 Biochemical Role of Vitamin B12 (Cyanocobalamin)
Biochemical Role of Vitamin B12 (Cyanocobalamin)
The various chemical forms of vitamin B12 described above are largely considered artifacts of isolation and processing. In biological systems, these forms are converted into active coenzyme B12 compounds, collectively known as cobamides.
These biologically active coenzymes do not retain groups such as CN or OH but instead function through specific cobalt-centered coordination chemistry.
 Active Coenzyme Forms
Active Coenzyme Forms
The two major physiologically active forms of vitamin B12 are:
- Methylcobalamin (active in the cytosol)
- Adenosylcobalamin (active in mitochondria)
Adenosylcobalamin is also known as 5’-deoxyadenosylcobalamin, and both methyl- and adenosyl- derivatives represent the primary functional coenzyme systems of vitamin B12.
Additional biochemical classification describes three coenzyme-related forms based on variations in the benzimidazole component:
- DBC (Dimethylbenzimidazole cobalamin)
- BC (Benzimidazole cobalamin)
- AC (Adenyl cobalamin)
Among these, adenosylcobalamin is the predominant form stored in the human liver.
Another important functional derivative is methylcobalamin, in which a methyl group is directly bound to cobalt.
 Biochemical Reactions Involving Vitamin B12 Coenzymes
Biochemical Reactions Involving Vitamin B12 Coenzymes
Vitamin B12-dependent coenzymes participate in several essential metabolic processes:
- Transmethylation reactions
- Involved in the synthesis of:
- Thymine
- Methionine
- Choline
- Methyl transfer and molecular rearrangement reactions
- Unlike simple hydrogen transfer reactions, B12 primarily facilitates group transfer and structural rearrangements.
- Nucleotide metabolism
- Supports utilization of deoxyribonucleotides in DNA synthesis
- Metabolic consequence of deficiency
- In vitamin B12 deficiency, methylmalonic acid accumulates
- This occurs due to failure of conversion:
- methylmalonyl-CoA → succinyl-CoA
- As a result, methylmalonic acid is excreted in increased amounts in urine
 Major Enzyme Systems
Major Enzyme Systems
Vitamin B12 functions as a cofactor for two critical enzymes:
1. Methionine Synthase
- Catalyzes: homocysteine → methionine
- Requires methylcobalamin
- Establishes a crucial metabolic link between folate and vitamin B12 pathways
2. Methylmalonyl-CoA Mutase
- Catalyzes: methylmalonyl-CoA → succinyl-CoA
- Requires adenosylcobalamin
- Essential in the metabolism of:
- Odd-chain fatty acids
- Certain amino acids
 Folate Trap Concept
Folate Trap Concept
In vitamin B12 deficiency, folate metabolism becomes disrupted due to the “folate trap” phenomenon.
- Folate is trapped as methyl-tetrahydrofolate
- It cannot be converted into other active folate derivatives
- This leads to a functional folate deficiency
- The ultimate consequence is impaired DNA synthesis, contributing to megaloblastic anemia
 Clinical Features of Vitamin B12 Deficiency
Clinical Features of Vitamin B12 Deficiency
Deficiency of vitamin B12 produces a wide range of hematological, neurological, and systemic manifestations:
- Megaloblastic anemia
- Glossitis (inflamed tongue)
- Neurological symptoms due to demyelination
- Peripheral neuropathy
- Subacute combined degeneration of the spinal cord
- Memory impairment
- Psychiatric disturbances in severe or prolonged deficiency
A key clinical point is that neurological involvement is more characteristic of vitamin B12 deficiency than folate deficiency, making it an important diagnostic distinction.
 Additional Biochemical Roles
Additional Biochemical Roles
Beyond its primary enzymatic functions, vitamin B12 has been implicated in several other biological processes, including:
- Conversion of folic acid into active coenzyme forms
- Metabolism of several amino acids
- Metabolism of choline and cholesterol
- Participation in porphyrin biosynthesis
- Activation of amino acids for protein synthesis




